 
Note that the answer to equation (6) is always a positive value for the photon's wavelength. In this model, energy levels, E n, of hydrogen-like atoms can be determined as, Niels Bohr proposed a model of the atom that explained with startling accuracy, the appearance of the spectrum of hydrogen. An example would be singly ionized Helium, which is the lightest hydrogen-like atom, besides hydrogen. Hydrogen-like atoms are those atoms with only one electron remaining, regardless of the number of protons in the nucleus. 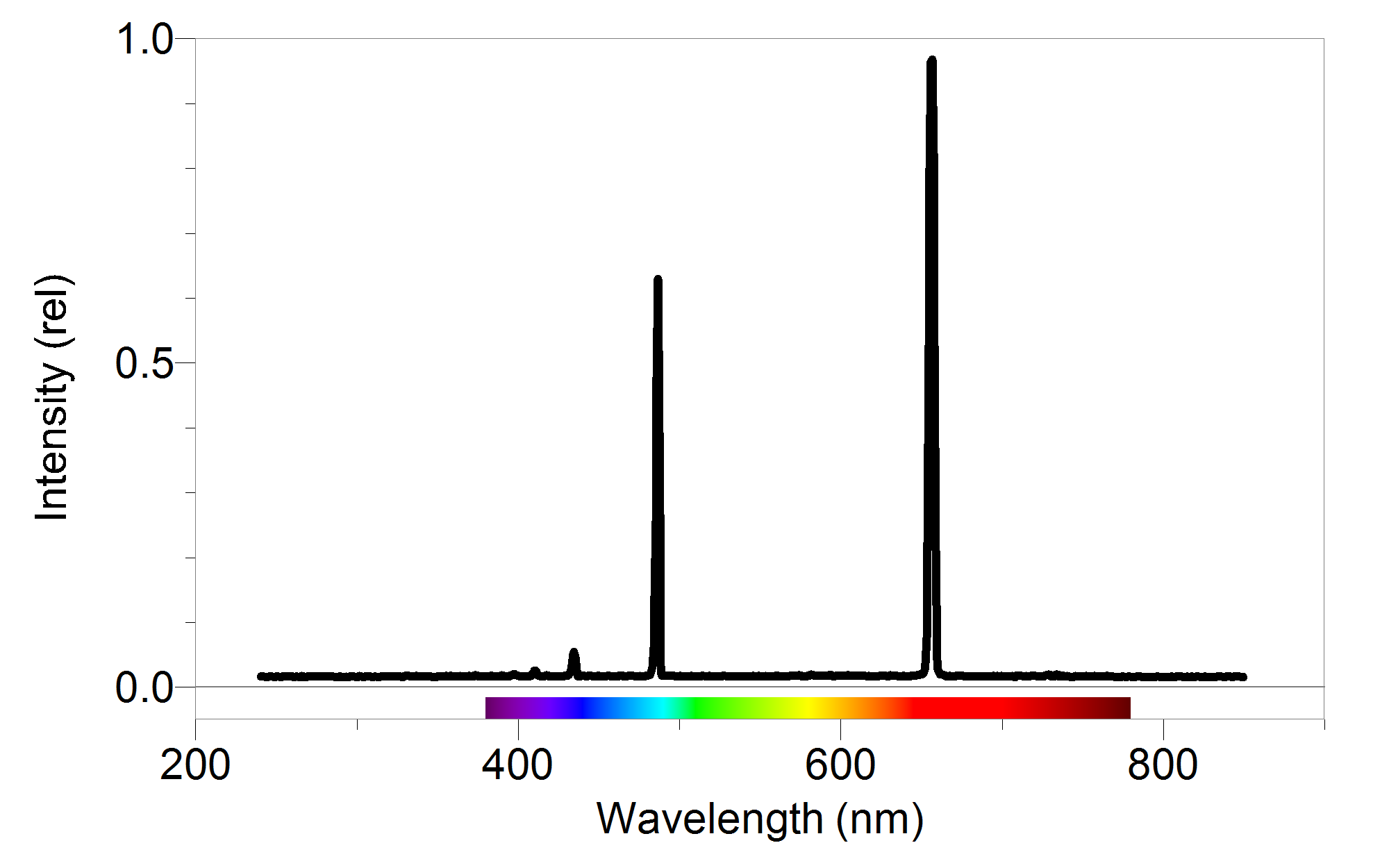
We see examples of this in the so-called emission nebulae, which are regions of rarified gas that are heated by stars off to one side of the nebula. Therefore the continuum source heats the object, and the electrons inside the atoms emit photons to move into lower energy states, which is always preferred by nature. Therefore we receive most of the light from the continuum source, except for those wavelengths that can promote electrons in the outer atmosphere to higher energy levels, thus removing these photons from the game.įor emission spectra, the source of the continuum is oblique to the line of sight between the observer and the object. Absorption spectra generally form when a continuum source, such as the central regions of a star, is directly in our line of sight, but behind our object of interest (which in this example), is the outer atmosphere of a star. Whether an object will present an absorption or emission spectrum depends greatly on the geometry of the continuum source with respect to the observer on earth. With absorption spectra we see essentially continuum emission with certain wavelengths of light missing and spectrographs usually render this as a black line.Īn emission spectrum on the other hand, shows little or no continuum emission, and only displays light at specific wavelengths. When looking at astrophysical objects we either see an absorption or emission spectrum. Matilsky discussed in his video lecture, atomic spectra occur due to the fact that orbital radii of electrons, and hence their energies, are quantized at specific levels determined by the atomic number (number of protons) and ionization state (number of electrons) in any given element. 
Analyzing the Universe - Course Wiki: Atomic Spectra Fingerprints of the Elements: Atomic SpectraĪs Dr. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
